



Epidemiological Report On The UK Outbreak
Issued by Defra. This is an initial Epidemiological Report On The Outbreak Of Bluetongue In East Anglia And South East England From Investigations Completed To 19 October 2007.
SUMMARY
- The current findings support the working hypothesis that infection was initially introduced into the counties of Norfolk, Suffolk and Essex on the night of 4/5 August by windborne transmission of infected midges from continental Europe.
- Investigations and analyses are in progress to determine whether or not there was a separate windborne introduction of midges into Kent and East Sussex. At the present time the cases of infection observed in these two counties are not explained by the movement of animals.
- Up to 19 October bluetongue virus (BTV) infection has been confirmed in 17 cattle herds and 8 sheep flocks as result of the reporting of suspect clinical cases of BTV. A further 22 cattle herds have been identified as infected but as yet clinically unaffected as a result of active surveillance in the vicinity of the first four detected clinical cases. The strain of the virus (BTV8) is the same as that present in northern Europe since 2006.
- The observed incidence of clinical disease within affected cattle herds and sheep has been very low with the majority of herds and flocks experiencing singleton cases. Two deaths have been observed in one sheep flock.
- The prevalence of infected herds in the vicinity (< 10km) of the four first cases of disease identified is currently 59%.
- The within herd prevalences of infection in cattle herds are generally low with an average just under 10%, but are lower in cattle herds in which clinical cases of BTV have not occurred. The within flock prevalence of infection in sheep is also very low at the present time with the majority of flocks having less than 3% infected as determined by serological testing for antibodies to the BTV.
- The expected greater prevalence of infection in cattle, compared with sheep, at the beginning of the infection process has been observed.
- The results re-inforce the findings in Northern Europe in that any active surveillance will involve sampling relatively large numbers of animals.
- Plans for further objective driven active surveillance are currently being considered.
INTRODUCTION
- Infection with Bluetongue virus (BTV) was confirmed on 22 September 2007 in a native born Highland cow on a farm, near Ipswich. This animal was initially reported as a suspect case of FMD by the attendant veterinary surgeon. The initial formal investigation by Animal Health veterinary staff ruled out FMD, but it was considered that the clinical signs could be due to BTV infection. Appropriate samples were taken for laboratory examination at the World Reference Laboratory, Institute for Animal Health (IAH), Pirbright. The virus was typed by the accepted molecular genetic methods as BTV8 (Professor Peter Mertens, IAH, Pirbright, personal communication.) Both serological and RT-PCR tests on samples from this initial case were positive. The results indicated that the clinically affected cow had been infected for 10 – 20 days (Dr. Chris Oura, IAH, Pirbright, personal communication.)
- The following provides the initial findings from the epidemiological investigations of the current outbreak up to 19 October.
ORIGIN OF INFECTION
- As indicated above, the virus isolated from the first case was identified as the BTV 8 strain, by colleagues at the World Reference Laboratory (WRL), IAH, Pirbright. This strain was first identified in Northern Europe (Belgium, The Netherlands, Germany, Luxembourg and France) in 2006 and has caused major outbreaks of disease in these countries in 2007.
- Since the identification of BTV infection in continental Europe there has been a ban on imports of susceptible animals from Restricted Zones in other countries.
- Until 4 August 2007, when the importation of live animals was prohibited under the Foot-and Mouth-Disease legislation, there had been post import testing of all susceptible livestock (and zoo animals at risk) from free areas of BTV affected EU Member States and from BTV free EU Member States (where a risk assessment supports this) within 10 days of arrival. Imported animals are maintained on their first premises of destination until negative results received, with advice to isolate from other susceptible animals where possible. The laboratory tests on all such animals tested have been negative to date.
- There has been considerable interest in the potential windborne transmission of infected Culicoides spp in Europe. In June 2006 Defra commissioned research by the Meteorological Office and the IAH, Pirbright to investigate the spread of BTV through the windborne Culicoides. One specific objective of this research was to develop an operational early warning scheme for Great Britain. As a result this operational service became available on 1 April and the model developed is run everyday to assess the likely risk of viable Culicoides reaching the GB coastal areas.
- An analysis of the daily outputs indicates that the most likely time that Culicoides could have been introduced to GB was overnight on 4/5 August (John Gloster and colleagues, Meteorological Office, personal communication).
- The area at risk from this airborne route, which is centred on East Anglia, is depicted in Figure 1.
- The epidemiological findings are consistent with such a relatively widespread introduction over a very short period of time (hours). The first reported cases in cattle, with clinical signs suggestive of bluetongue, were made within a four day period. These were in cattle herds in Lowestoft, in the county of Norfolk, Ipswich in Suffolk and Chelmsford in Essex
- The current working hypothesis is therefore that BTV8 was first introduced from Northern Europe on the night of 4/5 August as a result of the windborne carriage of infected Culicoides.
METHODS OF CASE FINDING
STATUTORY REPORTS OF SUSPECT CLINICAL CASES OF BLUETONGUE- Animals exhibiting signs suspicious or typical of bluetongue disease must be statutorily notified to the local Animal Health Division Office. Animals so reported are clinically examined by an Animal Health veterinary officer. If bluetongue cannot be ruled out on clinical grounds, samples are submitted to the IAH, Pirbright for examination by RT-PCR (for virus detection in blood samples) and serological testing. If either test method is positive bluetongue is officially confirmed.
- All susceptible species (ruminants) in the initial herds and flocks which are so confirmed with BTV infection are being re-sampled for serological testing to obtain estimate the prevalence of infection in the susceptible species on the premises.
- A retrospective desktop review of negated report cases for FMD during 2007 has been carried out to identify any potential reports that had clinical signs consistent with BTV. As of 19 October, six cases out of over 200 negated FMD suspect cases were identified with clinical signs consistent with BTV where no alternative diagnosis was recorded. (Of these six, one has been confirmed positive for BTV on serology, a second was negative and the remainder are under investigation. The details of these cases are not included in this report, but will be included in future reports.)
- Following the confirmation of BTV infection on the first 4 premises, 3 in the Ipswich area and 1 in the Lowestoft area (see Figure 1), a first stage active surveillance programme was instituted around these premises.
- In summary, this involved
- The testing of all cattle in all cattle herds within 3km of the three premises then (26 September) identified forming the Ipswich cluster and the one premises in the Lowestoft area.
- The testing of all cattle in cattle herds with more than 50 cattle in the area 3 – 10 km around the 3 premises forming the Ipswich cluster.
- The testing of all cattle in cattle herds in the area 3 – 5km area around the Lowestoft infected farm.
- The testing of all cattle in cattle herds with more than 50 cattle in the 5 – 10 km around the Lowestoft farm.
- The objective of this first stage active surveillance was to estimate the prevalence of infected cattle herds within 10 km of these initially observed infected premises. This together with the resulting estimates of the within herd prevalences of infection will aid the design of further surveillance.
CATTLE
- The most commonly recorded clinical signs observed in cattle have been: lethargy and depression, pyrexia (102.5-104F), conjunctivitis with increased lacrimation, serous nasal discharge, lesions of nasal mucosa (erosions/crusts around nares, peeling/sloughing of mucosa), oedema of lips, erosive lesions of lips and oral mucosa (with hyperaemia), increased salivation, stiffness/lameness, oedema of coronary band and lower limbs, hyperaemia and erosive lesions on teats.
- With the exception of three of the 17 premises from which clinical bluetongue disease was reported in cattle only one animal was affected at the time of the first visit. Of the other three premises, one herd had 2 clinical cases out of 147 cattle, one had 3 clinical cases out of 56 cattle and one herd had 4 clinical cases out of 47 cattle. No deaths attributable to bluetongue were observed in these herds.
- The most commonly recorded clinical signs observed in sheep have been: lethargy and depression, recumbency, respiratory difficulty, pyrexia (104-105F), oedema of face (especially lips), mucopurulent nasal discharge, increased salivation and hyperaemia of the nasal mucosa, and oedema of coronary band.
- With the exception of three of the eight premises from which clinical bluetongue disease was reported in sheep only one animal was affected at the time of the first visit. Two sheep were affected in a flock of 58. in another flock two sheep were affected in a group of 40 and in the other flock one sheep had died overnight and another sheep was in extremis, and therefore euthanased, having shown the same clinical signs. No other deaths attributable to bluetongue were observed in the other flocks. BTV was not confirmed in any goat herds or other ruminant species as a result of reporting suspect clinical cases of bluetongue. (One of two goats was serologically positive at the follow-up visit to a mixed farm in which one cow was clinically affected.)
- The above findings relate to the early course of infection and disease in these herds and flocks. Follow-up studies are therefore being planned in order to monitor the clinical incidence and the mortality rates in a sample, at least, of infected flocks and herds.
EPIDEMIOLOGICAL FINDINGS
REPORT CASES- The epidemic curves of the 153 report cases, in the period 21 September to 19 October, are shown in Figures 1a and 1b. BTV has been confirmed in 17 cattle herds and in 8 sheep flocks up to the 19 October as a result of the statutory reporting of suspect clinical cases of bluetongue.
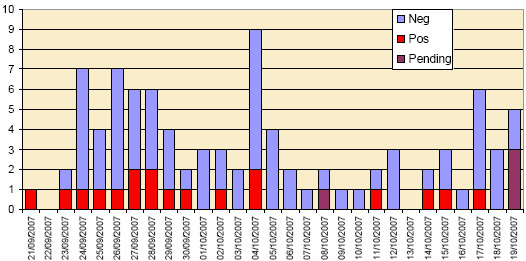
Figure 1a: “Epidemic curve” of report cases in cattle from 21 September to 19 October 2007
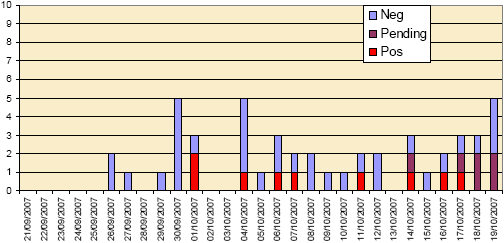
Figure 1b: “Epidemic curve” of report cases in sheep (and other small ruminants) from 21 September to 19 October 2007
GEOGRAPHICAL DISTRIBUTION OF REPORT CASES
- The geographical distribution of the 153 suspect cases of bluetongue reported from 21 September to 19 October in Great Britain is shown in Table 1. Suspect cases have been reported from 35 counties. Table 1: Distribution of all suspect cases of bluetongue reported since 21 September 2007, by county
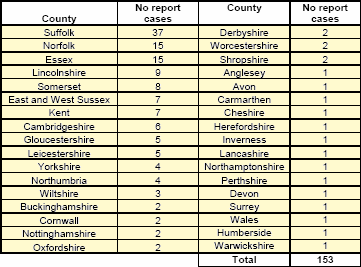
Table 1: Distribution of all suspect cases of bluetongue reported since 21 September 2007, by county
- The geographical distribution of the 23 confirmed cases detected by the statutory notifications is depicted in Figure 2. The distribution of these cases by county and the species infection was detected in is given in Table 2 Table 2: Distribution of reported clinical cases confirmed as bluetongue by county and species affected

Table 2: Distribution of reported clinical cases confirmed as bluetongue by county and species affected
- The relatively small number of report cases preclude any definitive conclusions, but this distribution has some notable features at the present time:
- The absence of confirmed report cases in sheep in Norfolk despite this county supporting a substantial sheep population (>100,000)
- The greatest observed incidences have been in cattle in Suffolk (~2% of herds) and in Essex (~1% herds)
- The absence of confirmed report cases in cattle in Kent, East Sussex and Cambridgeshire
- Until 14 October, the incidents of BTV infection were confined to three crudely defined geographical “clusters”: Ipswich, Lowestoft and Essex . Subsequently cases were detected in Kent and East Sussex and, perhaps more importantly in Peterborough, Cambridgeshire.
- The confirmed case of BTV infection in Peterborough was in one clinically affected ram on a farm comprising some 2,300 sheep and 20 cattle. It was a significant occurrence as it necessarily resulted in a new Control Zone and a considerable extension of the Protection Zone (Figure 2). This farm was subject to extensive sampling and laboratory testing which has resulted in no other susceptible species exhibiting any evidence of infection. The results of the investigations of this farm to date have not identified how infection reached this farm, except that introduction by infected animals on to the farm is extremely unlikely. These investigations are continuing.
- The recent occurrence of one case in sheep in Kent and of one case also in sheep in East Sussex, on the border of Kent and East Sussex, are the subject of current investigations. These include the possibility that these incidents are the result of a separate windborne incursion of infected midges, post dating the likely initial incursion on 4/5 August, from continental Europe. This is as opposed to the alternative hypothesis that this area was part of the original incursion, but it has taken a greater time to detect infection in the midge population as there is a relative deficiency of cattle to amplify virus production. Infection therefore only became apparent relatively later than in East Anglia following the relatively inefficient amplification in sheep.
CASES DETECTED BY ACTIVE SURVEILLANCE
- Twenty-two cases of BTV infection were confirmed in cattle herds as a result of the active surveillance in the Ipswich and Lowestoft areas (Figures 3a and 3b), a crude prevalence of 58.8% of the herds tested. There was no evidence for a statistically significant difference in the prevalence of infected herds in the two areas (Table 2).
- The prevalence of infected herds by herd size in the Ipswich and Lowestoft areas indicated an increasing risk of infection with increasing herd size Table 2. The results therefore indicate that active surveillance should be targeted at the larger herds.
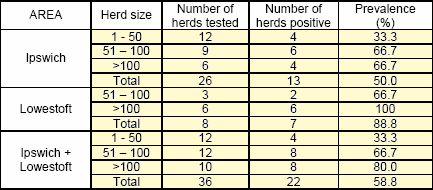
Table 3: Prevalence of BTV infected herds in the Ipswich and Lowestoft active surveillance areas, by herd size
WITHIN CATTLE HERD PREVALENCE
- In addition to the 22 infected cattle herds detected by active surveillance, twelve of the report case premises which have cattle have been subjected to follow-up surveillance involving the serological testing of all cattle. Table 3 indicates that the within herd prevalence was greater in these herds than in the herds detected by the active surveillance, in which all cattle were tested.

Table 4: Within herd prevalence of BTV infection, as assessed by serological testing, by method of case detection
- These results indicate that in the early stages of herd infection, at least, the prevalence of infection is relatively low. This has consequences on the sample size necessary to detect infected herds. Further case series studies are planned to monitor the dynamics of infection within herds, and particularly obtain estimates of the prevalence of infection as virus transmission wanes due to reduced midge activity.
- A small number of herds had a notably high prevalence of infection. The greatest prevalence (49.2%) was detected in a small suckler herd comprising 56 cattle. At present there are no obvious explanations for this prevalence of infection.
- There was no indication that the herd production type had an effect on the mean within herd prevalence of infection.
WITHIN FLOCK PREVALENCE
- Sheep, whole or part flock, have been tested on 7 farms in which BTV infection was confirmed. The results are summarised in Table 4.
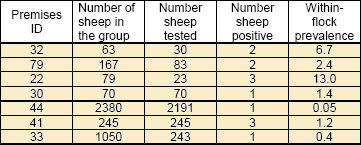
Table 5: Within flock prevalences of BTV in the seven flocks/part flocks tested by serology
- The within flock prevalences are notably low and generally lower than in cattle herds. These initial findings reflect the relatively short time since exposure to infected midges and the fact that sheep are the less attractive species compared with cattle.
OTHER SURVEILLANCE ACTIVITIES
- In order to gain an understanding of the geographical extent of BTV infection, animals moved from the Control Zones (see Figure 2) since 4 August 2007 are being traced and tested. The majority of these movements occurred during the short period 8 – 12 September when the FMD controls were relaxed, but licensed movements are also being traced. Priority is being given to the more distant movements. The results to date on the tests completed are negative, but a fuller description of the findings will be provided in further reports.
- The active surveillance in the Ipswich and Lowestoft areas is now complete. The objectives and therefore plans for future surveillance are currently being reviewed.
CAVEAT
- The results presented in this initial report relate to single points in time at the beginning of the infection process within flocks and herds and the population of the species at risk.. Also there is uncertainty of the awareness of the disease by animal keepers to determine the efficacy of the reporting of suspect cases of blue tongue. Appropriate care therefore needs to taken in interpreting these findings.
ACNOWLEDGEMENTS
We would like to thank Professor Peter Mertens and Dr Chris Oura and their staff at IAH, Pirbright for the molecular genetic investigations and the virological and serological laboratory testing and to John Gloster and colleagues at the Meteorological Office for the analyses of possible windborne transmission of midges. We are also indebted to colleagues in Animal Health for the collection of samples.
October 2007Further Reading
|
|
- You can view the full report, including graphs that were omitted from this article, by clicking here. |


